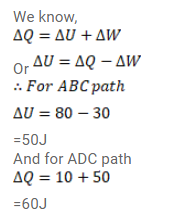Question:
When a system is taken through the process abc shown in figure (26-E3), $80 \mathrm{~J}$ of heat is absorbed by the system and $30 \mathrm{~J}$ of work is done by it. If the system does $10 \mathrm{~J}$ of work during the process adc, how much heat flows into it during the process?
Solution: