The terminology of different parts of the electromagnetic spectrum is given in the text. Use the formula E = hν (for energy of a quantum of radiation: photon) and obtain the photon energy in units of eV for different parts of the electromagnetic spectrum. In what way are the different scales of photon energies that you obtain related to the sources of electromagnetic radiation?
Energy of a photon is given as:
$E=h v=\frac{h c}{\lambda}$
Where,
h = Planck’s constant = 6.6 × 10−34 Js
c = Speed of light = 3 × 108 m/s
λ = Wavelength of radiation
$\therefore E=\frac{6.6 \times 10^{-34} \times 3 \times 10^{8}}{\lambda}=\frac{19.8 \times 10^{-26}}{\lambda} \mathrm{J}$
$=\frac{19.8 \times 10^{-26}}{\lambda \times 1.6 \times 10^{-19}}=\frac{12.375 \times 10^{-7}}{\lambda} \mathrm{eV}$
The given table lists the photon energies for different parts of an electromagnetic spectrum for differentλ.
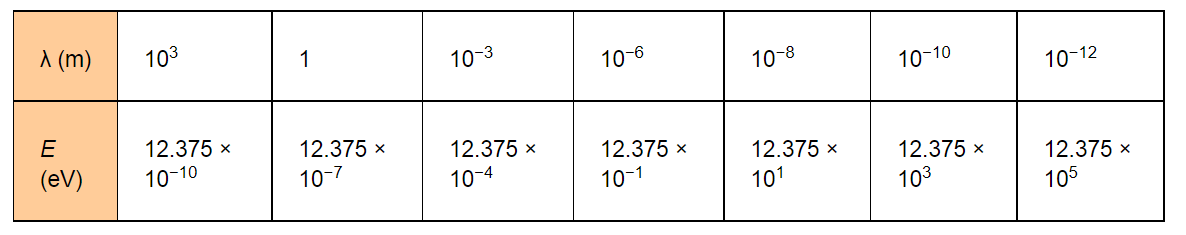
The photon energies for the different parts of the spectrum of a source indicate the spacing of the relevant energy levels of the source.
